Enzymes are drivers in living things. The most important thing about enzymes is that they are made of proteins that are chemically active and can speed up chemical reactions in cells without changing the processes themselves. As proteins, they are always made up of chains of amino acids with unique forms and links.
Enzymes can make cellular processes go faster and better. They are different for each type of answer. Without enzymes, the process moves along very slowly. Because of this, it can’t happen without an enzyme.
Structure of Enzyme
Enzyme have the following components:
Globular Structure
There are a lot of different amino acids that make up enzymes. When these amino acids come together, they make a round shape.
Active Site
The enzyme’s catalytic action is only in a small area called the “active site.” It has some amino acids in it. The enzyme’s round shape is made up of all the other amino acids. This active site takes part in a process called catalysis. The name for these chemicals is “substrates.” Attached to the active spot is the substrate.
An enzyme’s active site is made up of two parts:
- Binding site: It finds a certain substrate and combines with it to make an enzyme-substrate complex. The active site is turned on by this process.
- Catalytic site: The substrate is changed into a product at the active catalytic site.
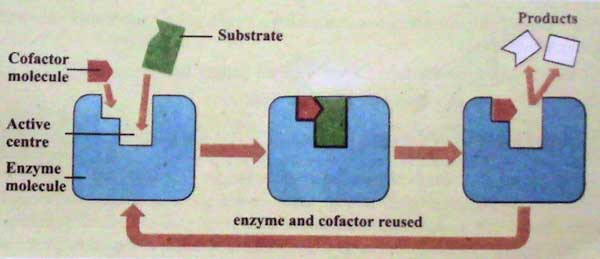
Co-factors
Cofactors are parts of enzymes that are not made of protein. Cofactors are necessary for these enzymes to work as they should. Cofactors are necessary for these enzymes to work as they should.
Cofactors help the enzyme connect to the substrate. Often, they are right in the middle of chemical processes. Cofactors can sometimes be used as a source of energy in chemistry. For a response to start, this energy is needed. There are three things that back this up:
- Activator: The detachable cofactor is called an activator. These cofactors are inorganic ions, such as Mg + 2, Fe + 2, Cu + 2
- Prosthetic group: The non-protein portion of the covalent is called a covalently bonded prosthetic group.
- Coenzyme: The non-protein portion that is loosely attached to an enzyme is called a coenzyme. Coenzymes are organic in nature but not protein. They are related to vitamins. Vitamins are the raw material for the synthesis of coenzyme. The coenzyme will not be destroyed during the reaction. Therefore, they can be used repeatedly. Therefore, less amount of vitamin is required in cells.
Apoenzymes
Enzymes with removed coenzyme or synthetic groups are called apoenzymes. Epoenzyme is still inactive.
Holoenzymes
The active enzyme composed of polypeptide chains and cofactors is called Holoenzyme. When cofactors combine with apoenzymes, holoenzyme are formed. This activates the enzyme and initiates the reaction.
Nomenclature
To speed up a certain kind of process, an enzyme will only work with one type of substance or group of substances. This is called the substrate. Because of this, enzymes are often called by adding “-ase” to the name of the substance they break down. For example, urease is an enzyme that helps break down urea. Not all enzymes have been named this way, though. To make enzyme names less confusing, a classification system has been made based on the type of chemical process the enzyme speeds up.
Characteristics of Enzymes
Enzymes are biological catalysts. They have the following characteristics:
- All enzymes are spherical proteins.
- They make you move faster. But they weren’t used to fight back.
- They don’t change what the end result is or how it works.
- Many target molecules can be changed by a small amount of the enzyme.
- Enzymes have a very clear job to do. Only one substrate or a group of linked substrates can be broken down by a single enzyme.
- They can be affected by changes in pH, temperature, and the amount of material.
- Some enzymes can’t do their normal jobs without cofactors.
- They lower the release energy. The activation energy is the least amount of energy needed to start a process.
- Some enzymes can do damage if they are turned on in the wrong place. For example, pepsin is an enzyme that helps break down proteins. It can damage the way cells are built from the inside. So, it is made in a form that doesn’t work called pepsinogen. Cells make enzymes and connect them to lysosomes. Lysosomes are bodies with membranes.
- In the chemistry process, they are not used up. After being used, they break apart so they can be used again.
- Some enzymes also have a part that is not made of protein. This part is called a prosthetic group.
- In certain biological processes, enzymes are used. Not every process needs every enzyme.
- They can combine with both acidic and alkaline things because they have proteins in them.
- Some ions or salts may turn on the action of enzymes. These ions or salts, like Ni, Mn, Mg, and Cl, are called activators. They lower the release energy.
Amino Acids Structures Chart and Names