Lipid are heterogeneous compounds related to fatty acids, which are insoluble in water, but soluble in organic solvents such as ethanol, ether, chloroform, and benzene. Lipids include fats, oils, waxes, cholesterol, and related compounds.
Fats, oils, and steroids form lipids. These compounds are made up of hydrogen, oxygen and carbon, but the amount of oxygen is small. Lipids are insoluble and have a low density in water.
Its include wax, natural rubber, vitamins A, E and K.

David Bloor coined the term “lipid” in 1943. Lipids can be defined as a group of organic compounds, they are insoluble in water, soluble in ether and ethanol (ratio 2: 1) of Bloor or other fat solutions (eg benzene, acetone, chloroform, and ether). In the mixture of. Lipids from fatty acids when they are hydrolyzed.
Lipids are found in both plants and animals. In plants, they are commonly found in seeds, fruits, and nuts.
Structure of lipid
The structure and properties of two representative lipids. Both stearic acid (a fatty acid) and phosphatidylcholine (a phospholipid) are composed of chemical groups that form a polar “head” and a non-polar “tail”. Polar heads are hydrophilic or soluble in water, while non-polar heads are hydrophobic or insoluble in water. Lipid molecules of this composition spontaneously form mixed structures, such as micelles and lipid bilayers, the hydrophilic end of which faces the aqueous medium, while the hydrophobic end is shielded by water.
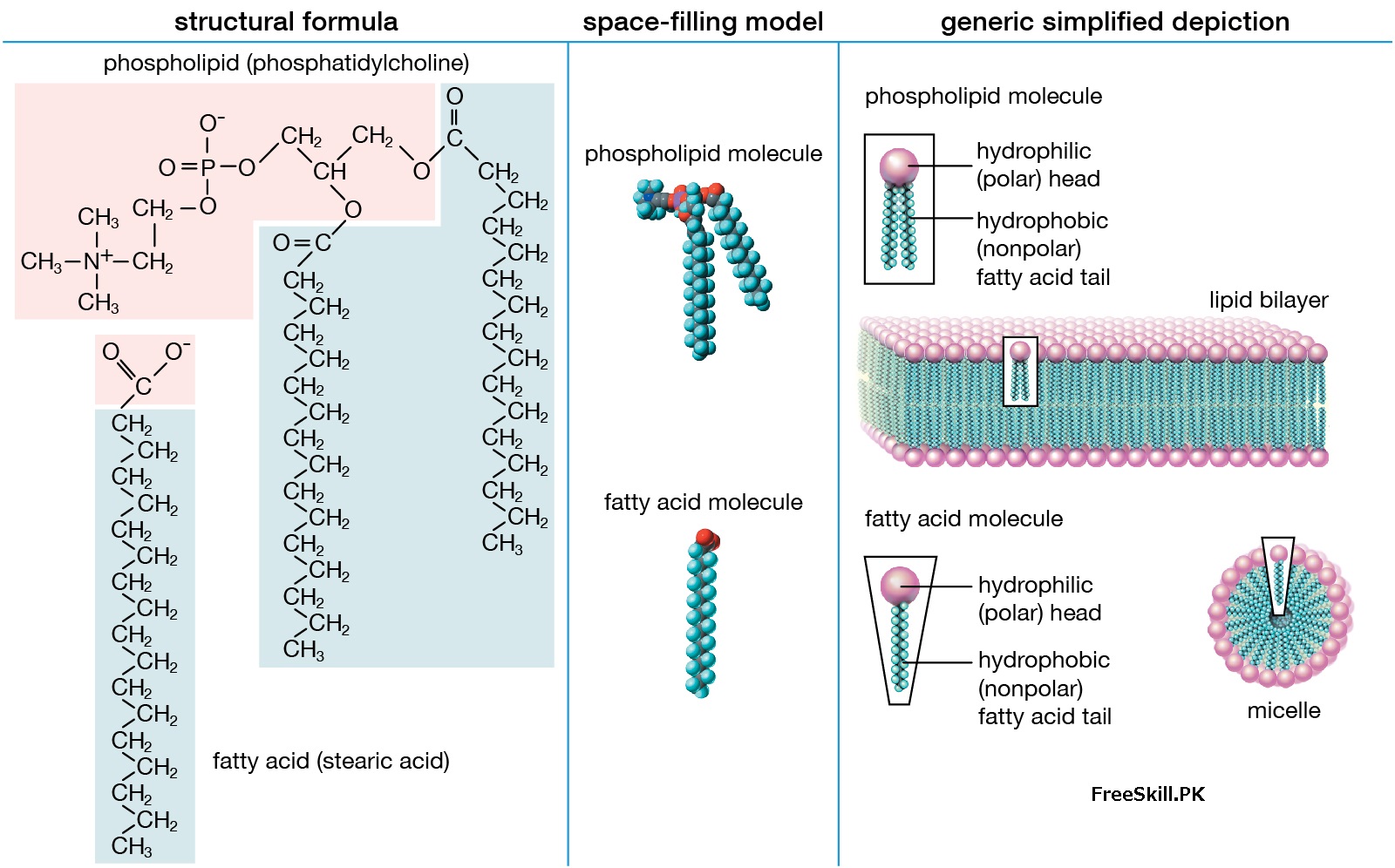
Lipid Function
Lipids perform the following functions:
- Lipids are hydrophobic compounds. Therefore, they form a component of the cell membrane.
- Lipids act like fats to store compounds.
- They have a high ratio of C – H bonds and a low ratio of oxygen. Therefore, compared with carbohydrates, lipids can store twice as much energy.
- Some lipids provide the ability to separate atmospheric heat and cold. They also act as waterproof materials. For example, wax in the insect exoskeleton. The waxy keratin forms an additional protective layer on the epidermis of some plant organs, such as leaves, fruits, and seeds.
- Some steroid hormones, cholesterol, and other lipids play important roles in animals.
Example of Lipids
Lipids divided into the following groups:
- Acyl-Glycerol (Fats and Oils)
- Waxes
- Phospholipids
- Terpenoids
1. Acyl-Glycerol (Fats and oils)
Fatty acids and alcohol esters called Acylglycerols. Esters compounds formed by the chemical reaction between alcohol and acid. During this reaction, water molecules released. The most abundant acyl glycerol as triacylglycerol. Also called triglyceride or neutral lipid. It contains three fatty acids.
Formula
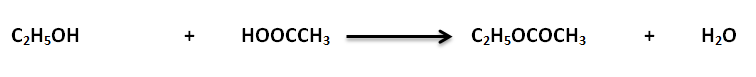
OH-is released from alcohol, and H + is released from fatty acids. H and OH together form water molecules. Acylglycerols make fat in plants and animals.
WAXES

A mixture of alkenes (C25 to C35), alcohols, ketones, and long-chain fatty acid esters called waxes, such as keratin. They perform various functions in animals and plants: wax
In plants: They form a protective coating on fruits and leaves. Wax can protect plants from water damage and abrasive damage.
In animals: Some insects secrete wax. They also provide a waterproof layer for insects, birds, and sheep.
Waxes are esters of fatty acids that contain longer monohydric alcohols or cholesterol than alcohol. They found in stems, stems, leaves, and fruit coatings, used in cosmetics, creams, ointments, and polishes.
Wax used as a machine lubricant. In the early days, the wax was obtained from whales. However, it is now obtained from a desert plant called Simmondsia Chinensis or Jojoba. It is rich in wax esters as lipids in seeds. CH3 (CH2) 4 COO (CH2) 29 CH3.
Phospholipids
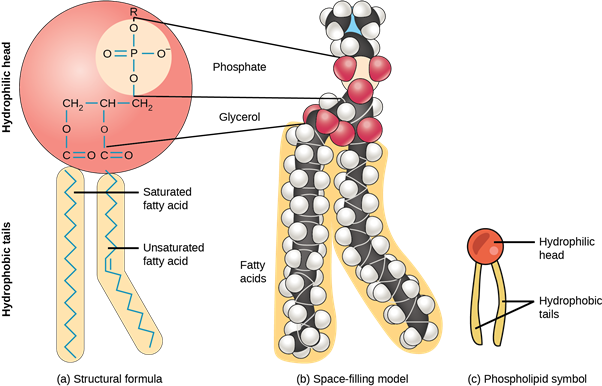
Lipids derived from fusidic acid are called phospholipids. Phosphatic acid composed of glycerol, fatty acids, and phosphoric acid. Nitrogen groups such as choline, ethanolamine, and serine are important components of phospholipids. Phospholipids are found in bacteria, animal, and plant cells. They form part of the cell membrane. Phosphatidylcholine is the most common cell membrane phospholipid. Phospholipid in lipids
They help in regulating cell permeability and transport processes. Phospholipids are actually complex lipids because the phosphate group replaces a fatty acid of lipids. Some other complex lipids are glycolipid and sphingolipid.
Glycolipids: These lipids associated with sugar molecules. When hydrolyzed, they will get compounds containing sugars, fatty acids, and nitrogen. They re found in chloroplasts.
Terpenoids
Lipids made up of simple repeating units of isoprenoids called terpenes. Different types of compounds formed by condensation of this unit. Terpenoids are very large and important compounds. For example, rubber, carotenoids, steroids, terpenes, etc.
They large lipid compounds made up of isoprenoid units (C5H8 units). Important categories of terpenoids are:
- Terpenes
- Steroids
- Carotenoid
1. Terpenes
The lipid group is based only on the isoprenoid units terpene (C5H8). Terpenes an important class of lipids derived from the steam distillation of plants.
Some of these terpenes are essential oils that cause many plant odors, such as rose, peppermint, camphor, cloves, sandalwood, eucalyptus, citrus, basil (Niazbo), etc.
2. Steroids
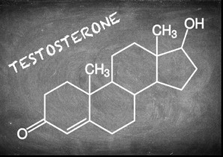
These are fats and will not make soap with NaOH or KOH. These consist of three six-member rings A, B, and C and one five-member ring. This structure called perhydro-cyclopentane-phenanthrene. These rings connected to a total of 17 carbon atoms and are called steroid cores.
Common examples of steroid lipids are cholesterol, vitamin D, and cortisone. Cholesterol found in animal tissues, nervous tissues, and spinal cord. It is also present in egg yolk and various fats. It is made in the liver. Steroids contain sex hormones, vitamins, and steroid hormones (such as cortisone).
3. Carotenoids

Carotenoids also included in this group. These are pigments that produce red, orange, yellow, and brown in plants. Vitamin A derivative of carotene and found in carrots, tomatoes, and leaves. Red carotene and yellow lutein are present in many plants. Tetrahydropyrrole is present in the pigment chlorophyll and cytochrome.
See Also:
- RNA: Describe its Structure, Types, & Functions
- Amino Acids: Its Types, Structure, Classification, and Functions
- Arteries vs Veins: What Are the Main Differences?